Parkinson’s disease is often thought of as a disease that announces itself late, with tremors, slowed movement, and stiffness. But long before those symptoms appear, something much quieter may already be going wrong inside vulnerable neurons.
A study at NCBS offers a closer look at what some of the early events of Parkinson’s disease might be. Using fruit flies, the research team found that two subtle errors in separate cellular systems combine to place a specific set of dopamine-producing neurons under stress. The result is an early build-up of harmful oxidative molecules inside mitochondria, the cell’s powerhouses. This kind of hidden stress could be one of the earliest steps on the road to Parkinson’s disease.
Parkinson’s disease can arise in two broad ways. In a small number of people, it is familial, caused by inherited mutations in specific genes. But in most cases, it is sporadic, with no clear genetic culprit. That has made sporadic Parkinson’s much harder to understand. Scientists suspect that, rather than a single major defect, it often emerges when several smaller vulnerabilities pile up over time.
The research team focused on two important players inside neurons. One is Parkin, a protein that helps cells identify and delete damaged mitochondria before they become dangerous. When this quality-control system fails, unhealthy mitochondria can accumulate and place neurons at risk.
The second player is IP3R, a channel that helps release calcium from the endoplasmic reticulum. Calcium is not just important for bones or muscles. Inside neurons, it acts like a messenger, helping regulate important processes like signalling and energy production. “Earlier work had shown that IP3R is especially important in a small cluster of dopamine-producing neurons in the fruit fly brain that help sustain flight. ” says Mrudula Dileep, the co-author of the study. “That overlap gave us an idea: what if a mild weakness in the Parkin pathway and a mild weakness in calcium signalling do not simply add up, but amplify each other?”
To test this, Mrudula and team created fruit flies carrying just one faulty copy each of the parkin gene and the itpr gene, which encodes IP3R. On their own, flies with either single defect looked largely normal. But when both partial defects were present together, the effect turned significant. These flies developed worsening flight problems as they aged.
“In fruit flies, flight is a powerful way to read out neuronal health. Flying is an energy-hungry behaviour that depends on precise coordination between muscles and the nervous system. If key neurons are under stress, the fly’s ability to sustain flight begins to falter,” says Mrudula.
The research team traced the problem to a particular subset of dopamine producing neurons in the fly brain, known as PPL1 neurons. These neurons are important for maintaining flight. In the doubly affected flies, these neurons were not yet showing the kind of dramatic collapse often associated with late-stage neurodegeneration.
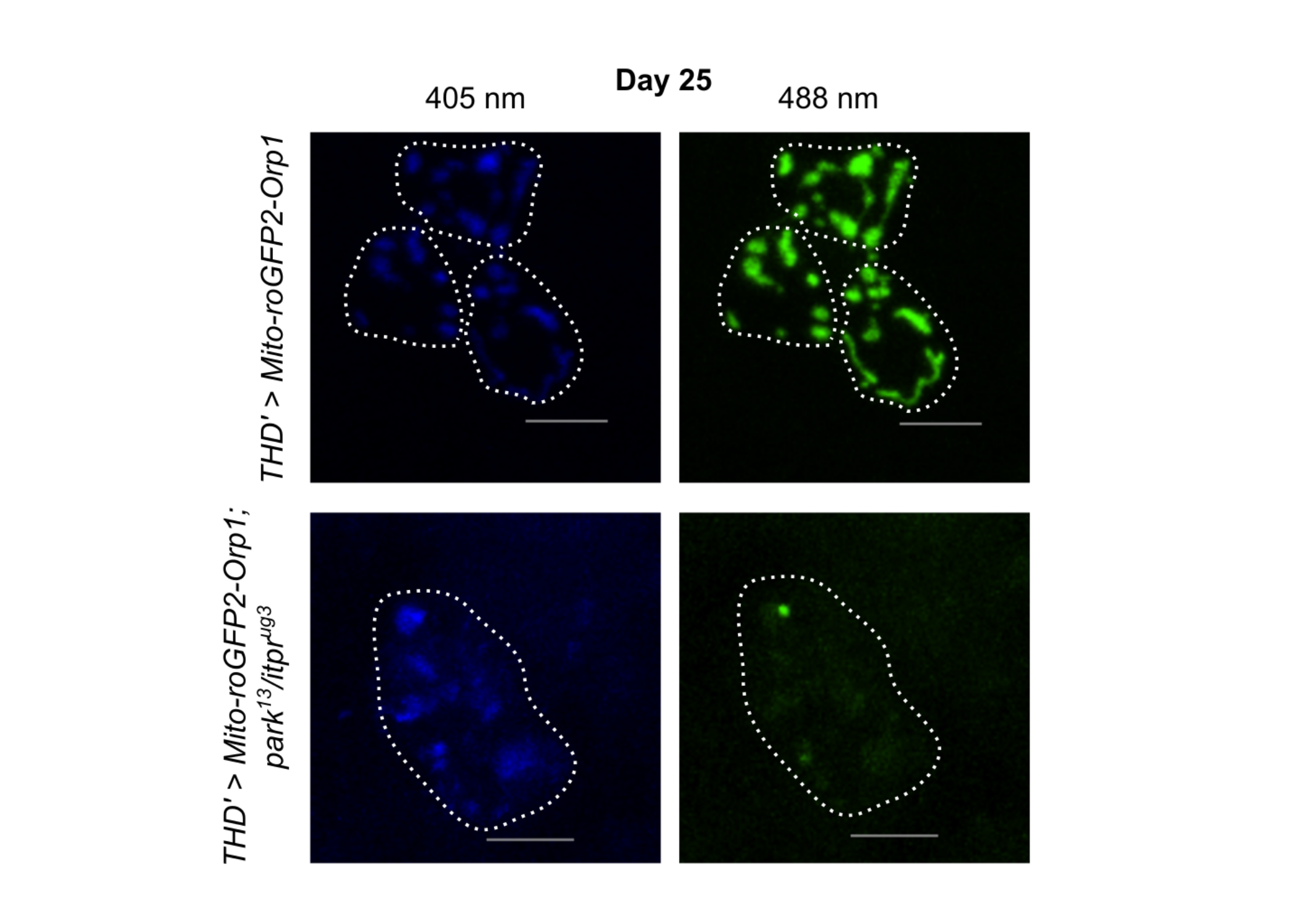
“We thought that we might find problems in mitochondria's handling of calcium or in mitophagy, the process by which damaged mitochondria are removed. After all, Parkin is deeply involved in mitochondrial quality control, and calcium strongly influences mitochondrial function,” says Prof Gaiti Hasan, the principal investigator of the study. “But that is not what we found. Mitochondrial calcium uptake appeared largely normal. Mitophagy was not strongly disrupted in a way that could explain the early defects either. Instead, the clearest signal was a rise in hydrogen peroxide, a reactive oxygen molecule, inside the mitochondria of these vulnerable dopamine neurons.” she added.
This means that the neurons seemed to be entering a state of oxidative stress before more dramatic failures appeared. Oxidative stress has long been linked to Parkinson’s disease, but it is often unclear whether it is a late consequence of damage or an early trigger. The researchers then investigated whether this oxidative stress is merely a sign of trouble or actually contributes to the problem.
To find out, they boosted the levels of catalase, an enzyme that breaks down hydrogen peroxide. When they did this in the affected dopamine neurons, the flies’ flight ability improved significantly. Lowering hydrogen peroxide levels also improved mitochondrial function. That suggests the excess hydrogen peroxide was part of what was driving the neuronal dysfunction.
The study also found another clue. The brains of the affected flies showed increased levels of DOPAC, a breakdown product of dopamine. This points to increased dopamine oxidation, a chemical process that can itself generate hydrogen peroxide. Dopamine is essential for neuronal signalling, but it is chemically reactive. In the wrong context, the very molecule that allows these neurons to do their job may also add to their burden.
“These findings help explain the high incidence of sporadic PD. Both genetic and/or environmental factors could together add to the oxidative stress of dopaminergic neurons, and contribute to cases of sporadic PD,” says Prof. Hasan






0 Comments