A novel regulator for membrane traffic in cells has been discovered by a team of scientists from the National Centre for Biological Sciences (NCBS), Tata Institute for Fundamental Research (TIFR) and the Babraham Institute. The molecule, Phosphatidylinositol 5-phosphate 4-kinase or PIP4K is a molecular overseer of membrane dynamics that functions as a negative regulator of endocytosis.
Keywords: membrane traffic, Phosphatidylinositol 5-phosphate 4-kinase (PIP4K), negative regulator, endocytosis
What do the Mumbai dabbawalas and the international e-tailer Amazon have in common with a living cell? The answer lies in their detailed organisation and ability to carry out complex operations – Logistics.
Within cells, the flow of information and goods between cellular compartments is tightly regulated by a logistics network that is incredibly intricate. And like all such networks, cellular logistics also require overseers – a set of molecular supervisors that direct the flow of materials between various locations in the cell for the smooth functioning of life.
Now, a team of researchers from the National Centre for Biological Sciences (NCBS), Bangalore, Tata Institute of Fundamental Research (TIFR), Mumbai and the Babraham Institute, UK, have identified a novel molecular overseer that regulates membrane traffic in cells. Using the fruitfly Drosophila as a model system, Raghu Padinjat from NCBS and his group have shown that the protein Phosphatidylinositol 5-phosphate 4-kinase or PIP4K, is essential in controlling membrane dynamics in cells.
The membranes of a cell are ever changing and never constant – being incessantly altered through endocytosis and exocytosis. Endocytosis occurs when parts of the cell’s surface membrane are internalized into structures called endosomes, while exocytosis is a process by which membranes from within the cell are sent to the surface.
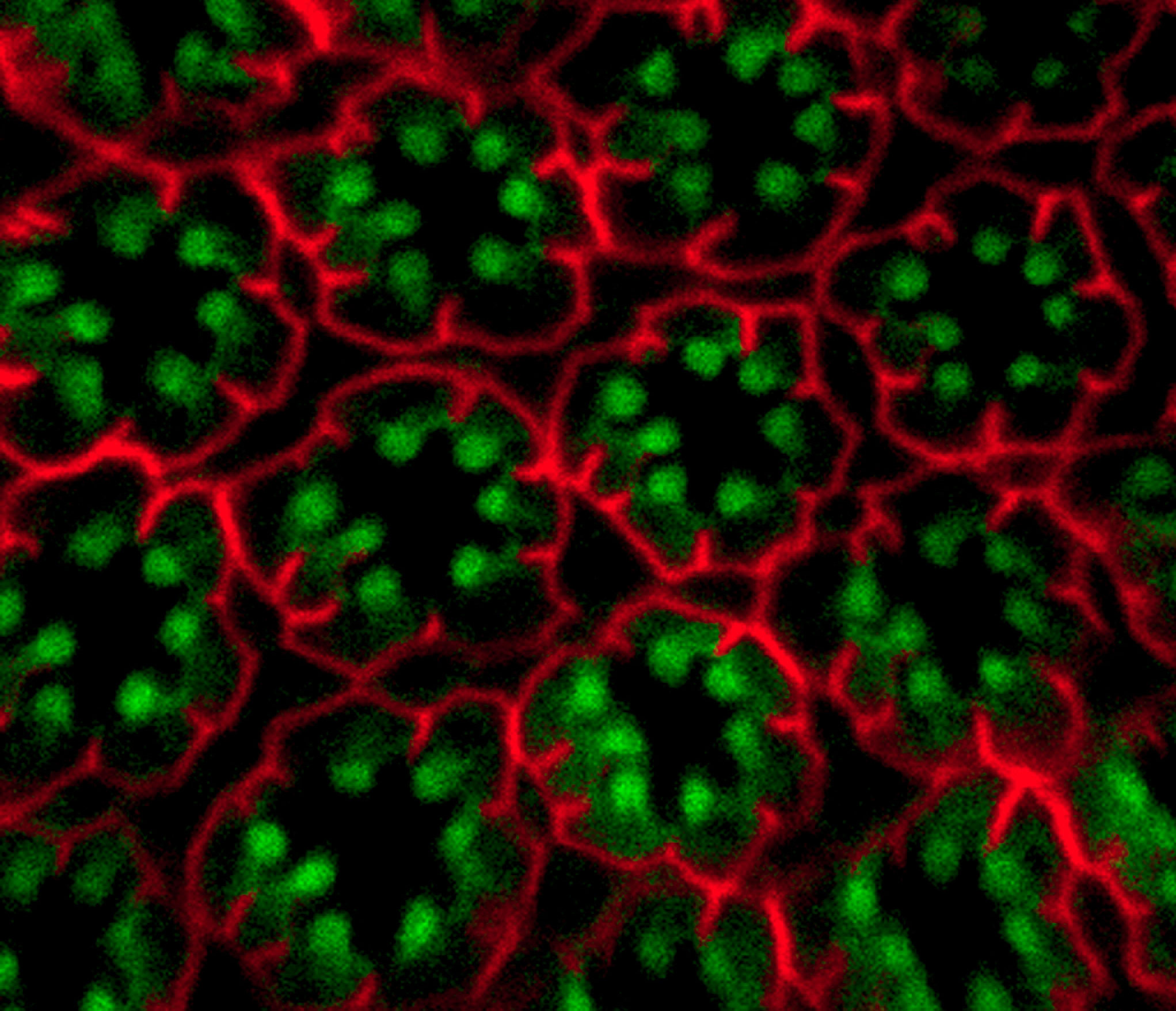
These processes are important in all cells including the retinal membranes of the eye, where the light-sensing molecule rhodopsin is anchored on photoreceptor cells. Rhodopsin, a membrane protein, is the receptor that absorbs light. Once triggered, rhodopsin must be engulfed into the cell through endocytosis, reset, then recycled back to the surface to maintain vision. However, when PIP4K is absent in photoreceptor cells, the rhodopsin that should be on surface membranes ends up inside the cell in endosomes.
While investigating this phenomenon, Padinjat’s team found that membrane traffic in cells without PIP4K seemed to be normal in all aspects, except one – endocytosis. When PIP4K is absent in photoreceptor cells, the process of endocytosis goes into overdrive and rhodopsin accumulates in expanded endocytic compartments inside the cell.
As Padinjat explains, “In real life logistics, if the flow of goods into a sorting centre does not match distribution from it, then parcels will build up at the sorting centre. This is what happens when PIP4K is missing; cell surface receptors enter and accumulate in the early endosomal compartment”. Furthermore, the role of PIP4K as a membrane traffic regulator is not restricted to photoreceptors alone, it also plays the same role in other cell types in Drosophila.
“PIP4K seems to be fine-tuning signals coming from the plasma membrane by modulating trafficking events – in this case endocytosis in cells. But what is really interesting, is that PIP4K seems to turn down the process of endocytosis,” says Kamalesh Kumari, a visiting scholar from TIFR, and lead author in the paper that describes these results. The novelty here lies in the fact that most known regulators of endocytosis are positive regulators – the loss of such molecules would decrease endocytic traffic. “By contrast PIP4K seems to be a negative regulator of endocytosis, which means that its presence dampens endocytosis; and this makes PIP4K a really unusual regulator of endocytosis” she adds.
In mammals, PIP4K is implicated in the development of cancer and autoimmune disease. “However, the challenge now is to connect complex phenomena such as cancer and autoimmunity to the defective cell and molecular processes that are part of the disease process,” says Padinjat. Though his team has established that PIP4K affects membrane traffic in many cell types in Drosophila, Padinjat now wants to determine if PIP4K in human cells also functions in a similar manner. “We also need to study the molecular mechanisms involved, and once we establish this, our medium-term goal will be to relate the function of the human protein in the context of human disease,” he says.








0 Comments