Bacteria routinely face stress from radiation, toxins, metabolic by-products and even antibiotics secreted by other bacteria in the environment, which can damage their DNA.
To cope with this constant threat of DNA damage, bacteria have evolved a variety of DNA damage response pathways that spot and repair the damaged DNA. A common response widely studied in bacteria is the ‘SOS response’, triggered by all sorts of DNA damage. However, some stresses might require a more specific arsenal for the bacteria to protect its genome—for instance, aberrant DNA methylation. The addition of methyl groups to the correct location on the DNA is crucial to optimal cell functioning, when gone haywire, it can cause mutations or cell death.
In a recent study, Anjana Badrinarayanan's lab at the National Centre for Biological Sciences has discovered Cada2, a transcription factor that switches ‘ON’ a DNA damage response specifically to aberrant DNA methylation. The study finds that all major groups of bacteria have Cada2-like proteins that regulate adaptive response, creating a memory in cells that helps them repair DNA faster the next time methylation damage occurs. The findings highlight the importance of these essential proteins for bacteria to survive methylation damage.
Most understanding of the methylation damage repair comes from E. coli Ada (EcAda) response. Many bacteria such as Caulobacter crescentus are known to lack an adaptive response altogether.
“We were looking at how bacteria like Caulobacter known to lack an adaptive response cope with methylation damage”, says Aditya, lead author of the study.
Aditya and team treated Caulobacter cells with different DNA damage-causing agents including methyl methane sulphonate (MMS) that can inflict unwanted methylation on bacterial DNA. Using RNA sequencing, the team found a universal set of genes that were expressed under all damage conditions, and another set specifically expressed due to exposure to methylation damage.
To find out, if these methylation-specific genes were independent of the SOS response pathway, the team deleted the recA gene that activates the SOS response. They compared the RNA-seq data from the cells with and without recA gene. They found that in the absence of recA only methylation-specific genes were induced, while the universal genes remained unexpressed.
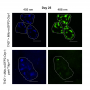
To dig deeper into the activity of genes involved in both pathways, the team put a fluorescent reporter under the regulation of the promoter of these genes– DNA sequences where the transcription of a gene(s) to mRNA is initiated. They took snapshots of these cells post-exposure to methylation damage at different intervals. They observed promoter activity of methylation-specific genes switch on after the SOS response has been initiated segregating both the responses in time.
The study finds that the methylation-specific response is also adaptive, implying that these genes can exhibit a swifter response if they had prior exposure to methylation damage-causing inducing agents.
“We wanted to look at what regulated the induction of this methylation-specific response”, says Aditya.
Aditya and the team zeroed in on three genes that were part of this Caulobacter adaptive response - cada1, cada2 and cada3. They knocked out each of these genes individually from the cells and exposed them to methylation damage. While cells in the absence of cada1 and cada3 cells could respond to the damage, the ones lacking cada2 could not initiate the adaptive response in Caulobacter.
Using computational approaches research team explored if proteins involved in adaptive responses were present across bacterial species.
The features of Cada2 are conserved across bacteria yet distinct from the structural and functional features of EcAda. Despite these differences, we observed similarities in the defining characteristics of the downstream response, adds Aditya. The findings highlight the evolution and divergence of adaptive response transcription factors in bacteria that drive essential bacterial DNA damage responses.









0 Comments