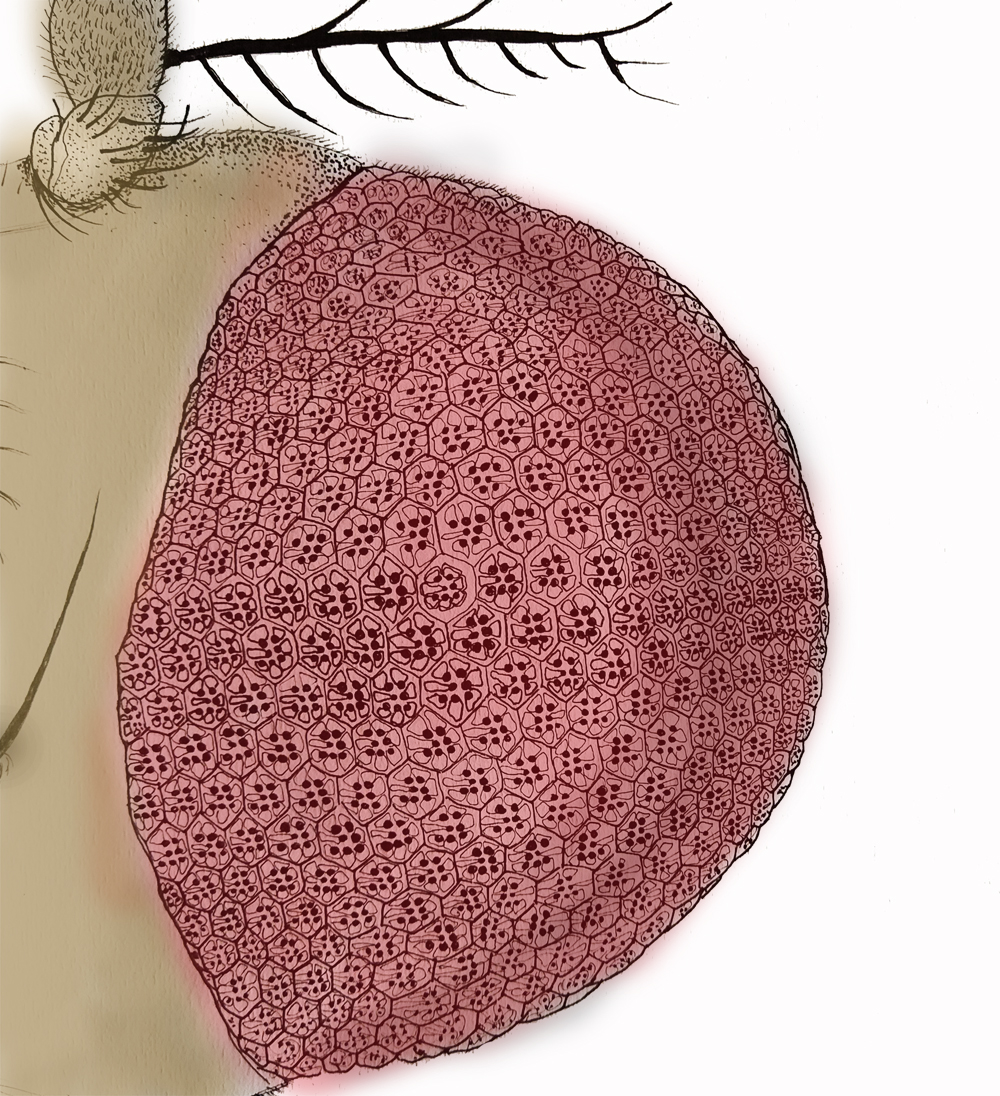
Image description: Line drawing of the Drosophila head showing the compound eye (pink). Individual repeating units of the compund eye are seen and individual cellular structures superimposed in each unit. Image credit: Dr. Deepti Trivedi.
Imagine diving into a eukaryotic cell.
You would be welcomed by a grandiose of controlled chaos – streamlined in time and space. But what would strike you most is the organization of the cell into functionally distinct partitions or organelles, each teeming with their unique set of proteins, lipids and other biomolecules. While this helps to compartmentalize cellular processes, the various organelles still need to network in order to sustain a harmonious cell. So, how do they do it?
For many years, communication between organelles was thought to be contactless (through packaged biomolecular signals), until a day came when scientists discovered membrane contact sites (MCS). To the untrained eye, MCS may appear like organelle membranes juxtaposed against each other. But, there is a miniscule space (ten thousand fold smaller than the width of your hair) between the two membranes. Numerous MCS are found along the pleats of the endoplasmic reticulum (ER) that brushes close to the plasma membrane (PM).
Since the discovery of MCS, scientists have been grappling with a number of questions. What holds MCS structure together (or apart) so that it doesn’t collapse the two membranes into one? What cellular processes take place at such close quarters? And importantly, what is the functional significance of MCS for an organism?
Thanks to a team led by Dr. Raghu Padinjat, we may now have some answers. In their recent study published in EMBO Reports, the researchers describe how a protein called extended synaptotagmin (Esyt) regulates the structure of MCS and also functions as a gatekeeper for lipid transfer across the ER-PM junction. The research, carried out in Drosophila photoreceptors, shows that Esyt helps localize a lipid ferrying protein (RDGB) at ER-PM contact sites and therefore averts retinal degeneration. This is the first study to highlight a role for an MCS localized Esyt in animal physiology.
“Esyts have been known for almost 10 years now. Earlier they were thought to function in the context of synaptic vesicle function and then their role in ER-PM contact sites was proposed. While their localization at ER-PM MCS could be clearly visualized in cultured cells, the requirement of Esyt in organismal physiology could never be established,” explains Padinjat.
One of the hurdles here was essentially a problem of too many. Most higher organisms have multiple copies of the Esyt gene in their genome. So when one is lost, the other compensates, thereby masking each other’s function. “Flies however, have a single gene for Esyt and hence this problem is resolved,” he elaborates.
Vaisaly, a PhD student in Padinjat’s lab and lead author of this study took upon the task of addressing this ‘gap’ in Esyt and MCS biology. She and her colleagues decided to carry out their experiments in Drosophila photoreceptors, where Esyt is found dotting the ER-PM contact sites.
To begin with, the team looked at flies that lacked the Esyt gene. These flies survived, but were developmentally delayed, smaller in size and lacked longevity. As ironic as it may sound, this was in fact great news. It meant that for the first time Esyt function could be visualised and quantified at the organismal level.
Next, when probing for Esyt function at the cellular scale, they found that flies devoid of Esyt lost the ER-PM contacts within photoreceptor cells. Surprisingly, the physiological properties of the photoreceptors were preserved, including their ability to relay light triggered messages (or photo transduction).
“It turned out that the cell can dispense the function of the protein,” says Vaisaly.
These data seem like Esyt is critical for shaping MCS at ER-PM junctions, but could be functionally done away with for photoreceptor function. But here comes the twist.
The group suspected that they could be missing out on Esyt’s cellular function if they ignored one of its indispensible neighbours - the retinal degeneration protein (RDGB). This suspicion encouraged Vaisaly to carry out genetic interaction studies between Esyt and RDGB.
RDGB ferries lipids across membranes, which is a vital step in photo transduction. Normally, flies mutant for RDGB show retinal degeneration. When Vaisaly challenged these RDGB mutant flies with a dearth of Esyt, they performed worse. In fact, subsequent experiments revealed that Esyt is what helps anchor RDGB at these contact sites so that lipid transfer across the ER-PM contact site is unruffled and photoreceptor integrity is maintained.
“Our paper is the first to show an in vivo function for Esyt. In fact, we can now consider reconstitution studies in flies that could help figure out how mammalian Esyts work!” exclaims Padinjat, who hopes that this study would show the way forward to understanding Esyt function in organisms.
Vaisaly is on the same page. She says, “The genetic interaction studies helped us to understand the importance of the protein in the context of in vivo cell physiology. It reaffirms our understanding that every event inside the cell is interconnected and well-coordinated to give a collective flawless output.” This is exactly where the study stands out – the fact that they decided to look at Esyt function – not in isolation, but as a socially interacting protein.
So, the next time you imagine diving into a eukaryotic cell, remember to pay heed to these lively inter-organelle straits that may appear too close for comfort. But in reality, are not.









0 Comments