More deadly than bullets and earthquakes, strokes claim millions of lives each year. A stroke occurs when an artery in the brain becomes blocked or leaks, often resulting in paralysis on one side of the body. Nearly 87% of stroke cases are caused by artery blockages, known as ischemia. Traditional surgical treatments to remove these blockages can lead to bleeding and damage to surrounding brain capillaries, potentially worsening a patient’s condition. Scientists are on the hunt for innovative and safe therapeutic strategies for treatments.
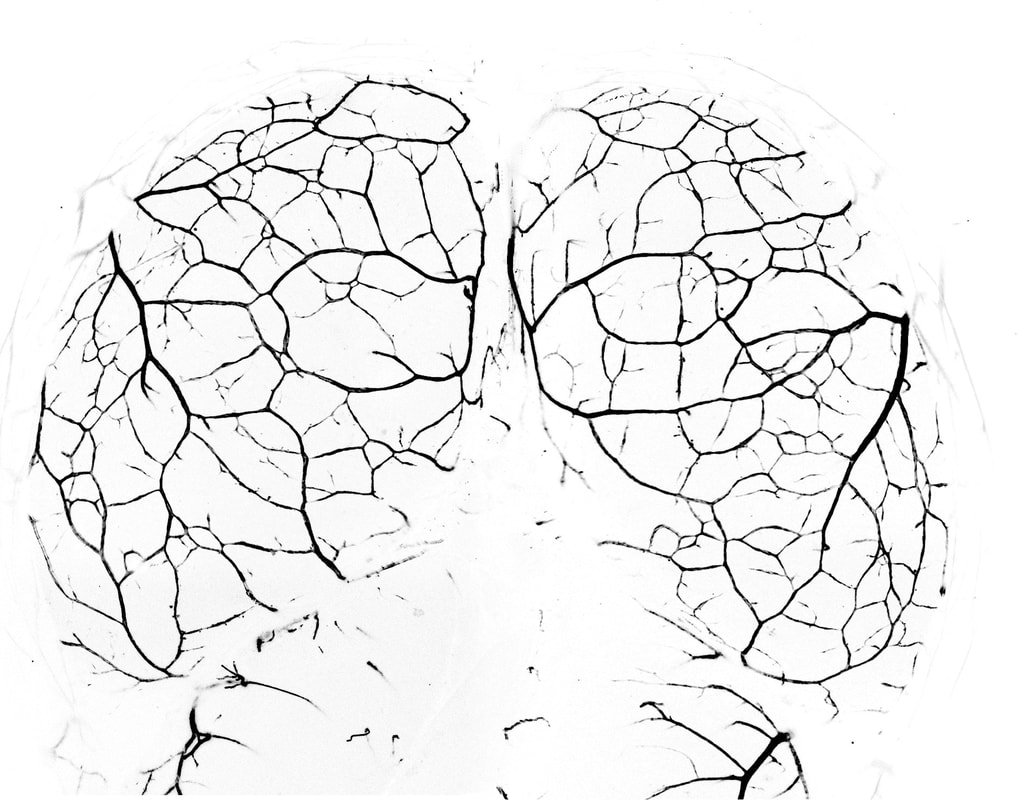
In a recent study, researchers at the National Center for Biological Sciences have found molecular clues that hold the potential to improve survival rates among stroke patients. Their findings could contribute to a groundbreaking treatment concept – redirecting blood flow away from blocked arteries towards healthy ones, offering hope for better treatment. Our brain relies on an intricate network of arteries for its blood supply. Doctors often assess the number of connections between these arteries, known as collateral arteries, to gauge a patient's survival chances. The more collateral arteries present, the greater the likelihood that blood can reroute from a blocked artery to a healthy one—much like diverting traffic from a jammed road to an open route. Building upon previous insights, scientists are looking at the genetic and molecular pathways that enable collateral arteries to mitigate the effects of blocked arteries. Understanding these processes is vital, as this life-saving capability is influenced by an individual's genetic makeup.
Collateral arteries are not unique to the brain; they also play a crucial role in other organs like the heart and intestines. A study published by Dr Soumyashree’s lab in 2023, looked at how collateral arteries grow in the heart. Their team found that endothelial cells - the building blocks of every blood vessel, bud off from the tips of arteries, expand in number and finally arrange themselves forming collateral arteries. To expand in number these artery endothelial cells de-differentiate and proliferate. Studies have suggested that this multi-step process requires at least two receptors on the surface of endothelial cells of arteries: VegfR2 and Cxcr4.
Following this, Dr Suraj Kumar and the team are now exploring processes involved in the formation of collateral arteries in the brain. In a recent study, they followed the family tree of artery endothelial cells by lineage tracing and observed the process of development of collateral arteries using confocal imaging in live mouse embryos. Their research indicates that collateral arteries in the brain form differently than in the heart. In the brain, the tips of the arteries grow towards each other until they connect. But how does one artery tip find the other? A trail of endothelial cells guides the way, but here is the twist: these cells do not originate from arteries. Instead, the capillary-derived endothelial cells lay down the tracks and pave the way for arterial growth to occur.
Kumar and team also found that while VegfR2 is crucial for initially forming collateral arteries in the brain, Cxcr4 plays no significant role. “Collateral arteries shrink and decrease in number as we age and this rarefaction makes us more susceptible to stroke and contributes to various neurodegenerative diseases like Alzheimer’s. Unlocking the reasons behind this vascular decline and finding ways to extend the lifespan of these vital arteries could have a profound impact on brain health in older adults, potentially enhancing their quality of life. With the world’s population ageing rapidly and healthcare costs rising, this line of research may hold the key to addressing some of the most pressing medical challenges of our time”, says Dr Suraj Kumar, the first author of the study.
The differing energy demands of the heart and brain may explain why collateral arteries form differently in these two organs in mice. Collateral arteries in the mouse heart only form in response to artery blockage, while in the brain, they develop during embryonic stages. This difference may also be attributed to the fact that neurons are more sensitive to low oxygen levels than heart muscle cells.
“We are looking at two completely different ways of making new arteries in two critical organs- the heart and the brain. We now have in hands, two fantastic preclinical models which can be used to carefully dissect out the various molecular cues that orchestrate each step of collateral formation. More than that, this gives us the opportunity to ask fundamental questions about what kind of signals (whether chemical or physical in nature) decide how a cell reacts to a given stimulus. Our lab continues to develop tools to pursue these very basic science questions. In the near future, we look forward to collaborating with clinicians and exploring the genetics underlying pre-existing collaterals in human patients” says Dr Soumyashree Das, the principal investigator of the study.









0 Comments