Rewiring the brain
As organs go, the brain probably has the most notorious reputation for complexity. Its millions of neurons are wired up into extremely precise circuitry within a very tiny space and yet this seemingly tangled mess is able to finely orchestrate the multitudes of exquisite behaviours that animals are capable of. It’s a wonder then, that despite this complexity, biology is able to unerringly reproduce this ‘mess with robust function’ in animals. How is this achieved during the development of the brain?
A recent paper in eLife addresses just this issue and describes an exciting and fundamental discovery about how complex brains are wired. In this paper, Sonia Sen and her collaborators, working on the brain of the fruitfly Drosophila melanogaster, show how a single gene, acting in a single stem cell controls the functional wiring of neural circuits in the brain.
The fly brain develops from 100 pairs of neural stem cells, each of which generates a family of related neurons. All neurons within a family, or ‘lineage’, tend to be very similar to each other - they look similar (have similar ‘projection patterns’) and operate in similar functional circuits. For example a given neural stem cell and its lineage might be wired into the taste circuitry, involved in the processing of taste information, while another might be wired into the olfactory circuitry, involved in processing olfactory information. How are these unique lineage identities established? It is thought that each neural stem cell must have a unique combination of genes, which in turn gives the lineage its unique circuit identities. Studies have shown that neural stem cells do have unique ‘signatures’ of gene expression. If these signatures do indeed translate to unique identities, might it be possible to change lineage identities simply by changing gene expression in the stem cell?
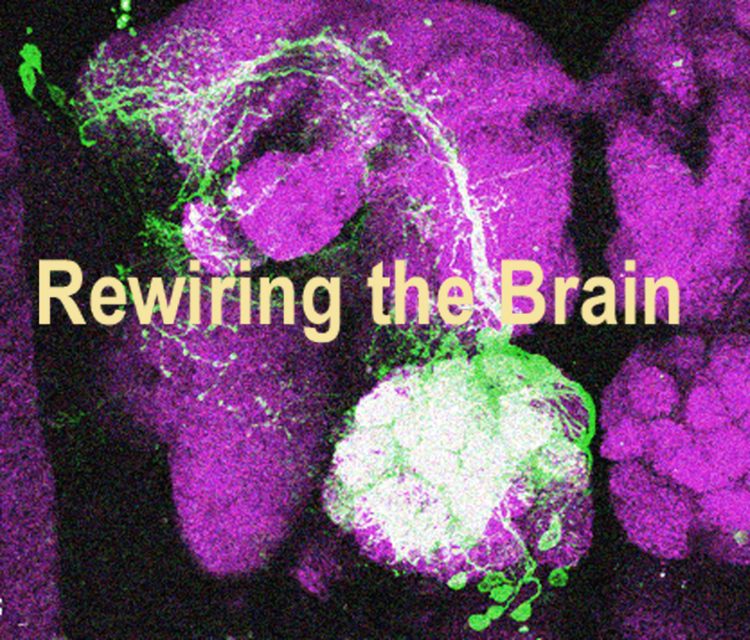
Sonia and her collaborators have done exactly this in the brain of the fly and thus provided evidence to support this long standing hypothesis in the field. They focused their study on two stem cells and their lineages in the brain - one which expresses the gene orthodenticle (otd) and another that does not. The neurons in these lineages wire into completely different neural circuits. While the otd positive lineage wires into a circuitry that is involved in sensorimotor control, the otd negative lineage wires into another circuitry that is involved in processing olfactory information. The authors artificially manipulated the expression of otd in the two stem cells and found that the presence or absence of otd determined whether the circuit identities of the lineage would be sensorimotor or olfactory.
They first removed otd from the stem cell that is normally otd positive and generates the sensorimotor neurons. In these experiments they found that the resultant neurons transform completely and resemble the olfactory identity neurons, which are normally generated from the otd negative stem cell. The projection pattern of these neurons transformed completely, as did their molecular properties, including the neurotransmitter identity. They even made synaptic connections with other neurons in the olfactory circuit. This extent of this neuroanatomical and molecular transformation was so dramatic that the authors then tested if the transformed neurons were at all capable of responding to odour stimuli. That is, could the transformed neurons actually function in olfactory information processing? Sonia went to Jing Wang’s lab at the University of California, San Diego and worked with Deshou Cao there to carry out some very tricky experiments to test the function of the transformed neurons. These experiments used functional imaging to monitor calcium activity (a read-out of neuronal activity) in the transformed neurons while they presented a variety of odours to antennae of the fly. If the transformed neurons were involved in processing olfactory information in the central brain, they would show neuronal activity in response to the odours being presented to the antenna. Deshou and Sonia found that indeed the transformed neurons showed strong neuronal activation in response to the different odours they tested. These results show that otd is a key gene in this stem cell and determines the sensorimotor (instead of olfactory) identity of its neurons.
The authors then asked if the converse might be true. Was the absence of otd from the olfactory stem cell necessary for it to generate olfactory identity neurons? Or would its abnormal presence in the (normally otd negative) olfactory stem cell transform its neurons into a sensorimotor identity? When the authors tested this by artificially expressing otd in the olfactory stem cell they found that indeed, the resultant neurons transformed to resemble the sensorimotor neurons. Thus, it was possible to completely change the identities of neural stem cells and their neural lineages in the brain by changing their molecular signature; most astoundingly, this was possible simply by changing the expression of a single gene in a single stem cell.
These novel findings have profound implications not only for understanding how neural circuits are assembled in the brain but also for the development of neural stem cell therapies, which are becoming increasingly important, and require the generation of very specific neuronal cell types. They also show a remarkable and unexpected plasticity of the brain to undergo large-scale rewiring fueled simply by changes in the expression of one gene in one stem cell. This almost trivial molecular change that can occur relatively easily in cells during development, may provide an elegant substrate for large-scale evolutionary modification of brain connectivity.
Sonia worked with Silvia Biagini and Ramveer Choudhary at the NCBS Bangalore where Heinrich Reichert from University of Basel is a frequent visitor to K. VijayRaghavan’s laboratory; and with Deshou Cao and Jing Wang at the University of California, San Diego for the functional imaging. The authors dedicated this work to their late friend and colleague, Veronica Rodrigues, whose legacy in olfactory biology continues to inspire.
Link to the eLife paper –
http://elifesciences.org/content/early/2014/12/24/eLife.04407
Comments
Post new comment