Gateways to plant cells and the route to salt tolerance
The endocytic process and its various roles have been extensively studied in animal systems. However, studies of these processes in plants have been limited and mostly focused on clathrin-dependent endocytosis. "In plants, the study of endocytosis is relatively nascent and the exploration in the field focused on endocytic uptake of individual candidate proteins- not on elucidation of the overall endocytic scenario that operates in a cell. As a result, we knew in detail about how individual proteins were endocytosed in a plant cell and the answer was almost exclusively clathrin-dependent endocytosis. We felt that other half, i.e clathrin-independent endocytosis was being grossly ignored and merited exploration", says lead author Anirban Baral, a researcher at MK Mathew's laboratory at the institute.
Seminal work by Satyajit Mayor, who is a co-author of this finding, was used as a 'stepping stone' in investigating clathrin-independent endocytosis in plants. The Mayor group had found that a unique clathrin-independent pathway for lipid-anchored protein uptake existed in animal cells.
Unlike animals, plants are sessile and often need to alter cellular processes in order to survive fluctuating environmental conditions. One of the greatest threats to plants, among other environmental hazards, is exposure to highly saline soil. This induces 'salt stress', a condition that affects the growth of the plant. The Mathew group and others have shown that one strategy to deplete the cytosol of excess Na+ is to sequester it in special compartments within cells.
However, such compartments are predictably of finite volume; only a limited amount of sodium can be stored. Therefore, there had to be a process whereby excess sodium could be removed. In light of this, Baral states, "Endocytosis and vesicular trafficking form an ideal candidate mechanism for flushing. We thought it would be interesting to explore how salt stress modulates endocytosis and how endocytosis in turn could be modulated to confer salt tolerance or sensitivity".
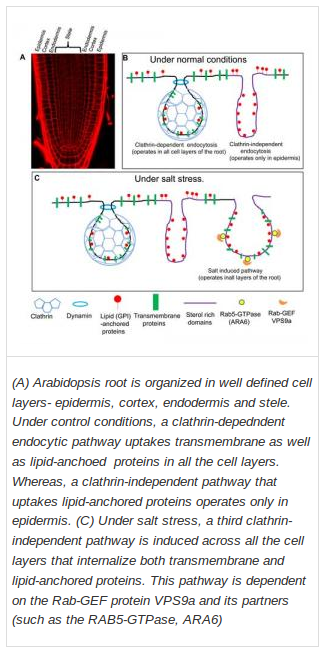
For the study, the researchers used the roots of Arabidopsis thaliana as a model. This system is ideal for observing endocytic pathways due to the transparency and simple and well-defined structure of Arabidopsis roots. Three classes of probes: lipophylic dyes, tagged transmembrane proteins and tagged lipid anchored proteins, were used to analyze endocytic processes.
The work was accomplished along with the support of Niloufer G. Irani (a former researcher in NCBS is currently with Department of Plant Sciences, University of Oxford, UK.) , Masaru Fujimoto ( Laboratory of Plant Molecular Genetics and Dept. of Biological Sciences, Graduate School of Agricultural and Life Sciences, University of Tokyo, Japan), and Akihiko Nakano (Dept. of Biological Sciences, Graduate School of Agricultural and Life Sciences, University of Tokyo, Japan' and Live Cell Molecular Imaging Research Team, RIKEN Center for Advanced Photonics, Wako, Saitama, Japan.)
"Mayor and I are long-time friends and when I was asked my opinion regarding Anirban's Ph.D. work at a meeting we both attended, I thought our live imaging expertise would be helpful. Mayor and Mathew gave Anirban an opportunity to stay in my laboratory in the University of Tokyo for a few months and Anirban worked well to learn knowhow of plant cell live imaging and collection of data. I am very happy to see that such an international collaboration worked very well leading to an excellent paper", said Akihiko Nakano
In agreement with previous studies, the recent work found that transmembrane proteins were taken up by cells mainly through clathrin-dependent endocytosis and that it operated in all cell layers. The study also demonstrated that clathrin-independent endocytosis functioned only in the epidermal (the outermost) layers and mostly in the uptake of lipid and lipid-anchored proteins.
The major finding of the study was the "different" endocytic process that kicked in under conditions of high salinity: an enhanced endocytosis and recycling mechanism that might help cells to 'flush out' excess sodium, and allow special compartments called vacuoles inside cells to expand in response to salt stress and store sodium ions. Under salt stress, there was a significant increase in the activity of both endocytic pathways. Absence of this mechanism would make the plant extremely sensitive to salt stress, leaving it unable to adapt to salinity.
"Of course, our search for a flushing mechanism is still on. This work builds a platform from which we can start to address that question. It took a great team of collaborators to get to this stage. The next step is (in typical fashion) a leap into the unknown - which makes it exciting for all of us." says MK Mathew
"With the changing climate and progressive salination of arable lands, salinity stress is a major threat to our food sustainability in future", said Anirban. "Understanding as many components of the salt adaptation machinery as possible, is important and will help in development of many more salt-tolerant crop varieties for the future".
The paper can be accessed at The Plant Cell: http://www.plantcell.org/content/27/4/1297.abstract?sid=d0e9fe00-4ed9-46c1-9cec-4fa242fabb11
Adrian Lyngdoh is a writer with the Research Media Services Division of Gubbi Labs.

Comments
Post new comment